 Other than CPS scores, OS remained significantly better with pembrolizumab in the total patient population: 13.0 months with pembrolizumab + chemotherapy versus 10.7 months with the EXTREME regimen.īecause toxicity is enhanced in chemotherapy-containing regimens, the hope was that pembrolizumab could hold its own as a single-agent versus chemotherapy + cetuximab, which it did. In most patients with a CPS ≥1, OS was also significantly better with pembrolizumab + chemotherapy versus EXTREME (13.6 vs 10.4 months P<.0001). In patients with a high CPS ≥20, pembrolizumab + chemotherapy led to a statistically significant improvement in OS, with a median OS of 14.7 versus 11.0 months with the EXTREME regimen ( P=.0004). Approximately 20% of patients across all arms had HPV-related disease. Most of the study patients (85%) had a CPS ≥1 and 43% had CPS ≥20. A total of 882 participants from 200 sites in 37 countries were randomized 1:1:1 to receive pembrolizumab alone (n=301), pembrolizumab + chemotherapy (n=281), or cetuximab + chemotherapy (n=300). 
Patients had known p16 status in the oropharynx and had provided tissue for PD-L1 testing. Patients were enrolled who had locally advanced metastatic HNC with good performance status (ECOG 0 or 1) and who were incurable by local therapies. The 3-arm trial compared pembrolizumab monotherapy versus pembrolizumab in combination with chemotherapy and the EXTREME regimen versus standard-of-care with the EXTREME regimen. The randomized open-label KEYNOTE-048 study demonstrated the superiority of pembrolizumab, alone or in combination, over the EXTREME regimen, 1 leading to the FDA approval of pembrolizumab + platinum-based chemotherapy and pembrolizumab monotherapy as new first-line standard-of-care therapies for the treatment of patients with recurrent/metastatic HNC. It’s important to incorporate this test into your routine practice, just as we now incorporate HPV testing in the oropharyngeal sites.” Additionally, both nivolumab and pembrolizumab are now FDA-approved for second-line treatment in recurrent/metastatic HNSCC. “In patients with CPS ≥1, you can start by giving single-agent-pembrolizumab, and omit the side effects and toxicities of chemotherapy in these patients. Haddad at the NCCN 2020 Virtual Annual Conference. “You are now required to get a combined positive score on your patient,” reported Dr. 2 Based on these results, the FDA approved pembrolizumab alone or in combination with chemotherapy as new first-line standard-of-care therapies for patients with recurrent/metastatic HNC squamous cell carcinoma (HNSCC). In the KEYNOTE-048 study, 1 patients on pembrolizumab demonstrated superior overall survival (OS) compared with those on the EXTREME regimen. Haddad, MD, Chief, Head and Neck Oncology Program, and Professor of Medicine, Dana-Farber Cancer Institute. Future exploratory analyses of tumor mutational burden and inflamed signatures could further evaluate predictors of benefit in patients with low PD-L1-expressing HNSCC.The biggest change to clinical practice for the first-line treatment of patients with metastatic head and neck cancer (HNC) is a new standard of care, according to Robert I. Analysis of the CPS <1 subgroup was limited by small patient numbers. In the CPS 1-19 subgroup, P+C vs E results were consistent with treatment benefit. Conclusions: There was overall evidence of increased efficacy with increasing PD-L1 expression. In the CPS 1-19 subgroup, HR (95% CI) for OS showed a slight advantage of P (n = 124) vs E (n =133) (0.86 ) and favored P+C (n = 116) vs E (n = 125) (0.71 ). Results: Baseline characteristics of the CPS <1, CPS 1-19, and CPS ≥20 subgroups were similar to those of the total population. 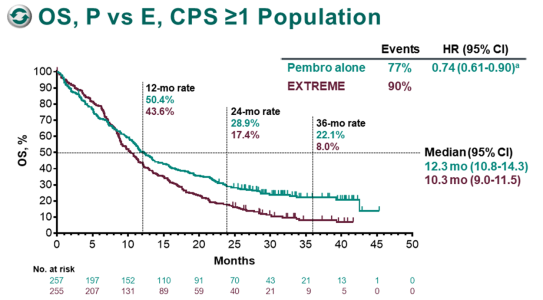
Hazard ratios and 95% CIs were based on a Cox regression model with Efron's method of tie handling, with treatment as a covariate. OS and PFS were estimated by the Kaplan-Meier method. Progression-free survival (PFS), objective response rate (ORR), and duration of response were assessed by RECIST v1.1 per blinded independent central review. Methods: Patients with R/M HNSCC and no prior systemic therapy for R/M disease were randomly assigned 1:1:1 to P, P+C, or E. Outcomes in CPS <1 and CPS 1-19 subgroups were not prospectively defined end points we present a post hoc analysis in these subgroups. Introduction: In the phase 3 KEYNOTE-048 trial (NCT02358031) in R/M HNSCC, first-line pembrolizumab (P) monotherapy vs EXTREME (E chemotherapy + cetuximab) improved overall survival (OS) in PD-L1 combined positive score (CPS) ≥20 and CPS ≥1 populations and led to noninferior OS in the total population with favorable safety first-line P+C vs E had superior OS in CPS ≥20, CPS ≥1, and total populations with comparable safety. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
